Example 1. Calculate the molar ratio [salt]/[acid] required to prepare an acetate buffer solution with
pH = 5.2.
Answer 1. Acetate buffer is composed from acetic acid (pKa=4.76) and its salt (e.g. sodium acetate). To make the calculation we use the buffer equation for acid- conjugate base pair. Accordingly, the solution to the above example is as the following:
Example 2. What is the pH at which [salt] = [acid] for an acetate buffer solution.
Answer 2. Given that [salt] = [acid] then [salt]/ [acid] = 1. The following describes the answer:
Example 3. Calculate the pH for a buffer solution composed from 0.01 M acetic acid (pKa = 4.76) and 0.02 M sodium acetate.
Answer 3. Using the buffer equation the answer is:
Example 4. Assume that an acetate buffer with a pH = 5.4 is needed such that the total buffer concentration ([salt] + [acid]) is equal to 0.05 M. Calculate the molar concentration for both acetic acid ([acid]) and sodium acetate ([salt]) required to make such a solution.
Answer 4. This example is solved in two steps. The first step involves finding the ratio [salt]/ [acid]. The second step involves solving two equations to find the required concentrations. The following describes the answer:
Step 1.
pH = 5.2.
Answer 1. Acetate buffer is composed from acetic acid (pKa=4.76) and its salt (e.g. sodium acetate). To make the calculation we use the buffer equation for acid- conjugate base pair. Accordingly, the solution to the above example is as the following:
Example 2. What is the pH at which [salt] = [acid] for an acetate buffer solution.
Answer 2. Given that [salt] = [acid] then [salt]/ [acid] = 1. The following describes the answer:
Example 3. Calculate the pH for a buffer solution composed from 0.01 M acetic acid (pKa = 4.76) and 0.02 M sodium acetate.
Answer 3. Using the buffer equation the answer is:
Example 4. Assume that an acetate buffer with a pH = 5.4 is needed such that the total buffer concentration ([salt] + [acid]) is equal to 0.05 M. Calculate the molar concentration for both acetic acid ([acid]) and sodium acetate ([salt]) required to make such a solution.
Answer 4. This example is solved in two steps. The first step involves finding the ratio [salt]/ [acid]. The second step involves solving two equations to find the required concentrations. The following describes the answer:
Step 1.
Step 2.
Example 5. What is the pH of a solution containing 0.09 M ephedrine (pKb = 4.64) and 0.01 M ephedrine hydrochloride (salt of ephedrine) given that pKw = 14.
Answer 5. To make the calculation we use the buffer equation for base- conjugate acid pair. Accordingly, the solution to the above example is as the following:
Feel free to comment or write questions.
MMGW5ZV9WDMF
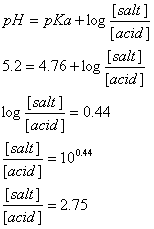


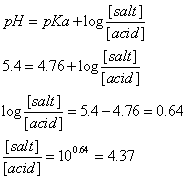


Thank you very much for this. Very well explained, clear and informative. Great work.
ReplyDeleteThis is proving very helpful for some biochem homework I have. One question though: in example 4, the 2nd step the first combined equation is:
ReplyDelete4.37×[acid]+[acid]=0.05
The next line is:
5.37×[acid]=0.05
Why does 4.37 become 5.37/where does this one come from?
Thanks
4.37×[acid]+[acid]=0.05 is the same as: [acid] × (4.37+ 1)=0.05
ReplyDeletewhich leads to the equation: 5.37×[acid]=0.05
I hope this helps.